Approach to Pancytopenia
Criteria for Pamcytopenia
Pancytopenia is a reduction in all three cellular components of the blood, namely, red blood cells (RBCs) and leucocytes (WBCs). The presentation is in the form of cytopenias leading to infections, anemia, or bleeding manifestations. In order to properly address every case of pancytopenia, there must be a thorough approach. so that it can be managed in the best possible manner.
It can be reduction in two cellular components (bicytopenia) or a reduction in all the three cellular components (pancytopenia).
In bicytopenia, anemia and thrombocytopenia (most common combination) leucopenia with thrombocytopenia (least common)
For practical purposes, Pancyotopenia Criteria
- haemoglobin <10g%,
- absolute neutrophil count < 1,500/cumm and
- platelets <1,00,000/ cumm.
Severe Pancytopenia
- It is labelled as severe when the three values are <7g%,
- <500/cu mm and
- <20,000/cu mm respectively.
Common Causes of Pancytopenia
Impaired production –
bone marrow failure disorders and marrow infiltration disorders
- Aplastic anemia – acquired and congenital
- Malignancy
- Primary and
autoimmune
myelofibrosis - Granulomatous
disorders - Metabolic disorders
Peripheral destruction
- Autoimmune Hemolytic Pancytopenia
- Splenic Sequestration
Impaired production and peripheral destruction
- Paroxysmal Nocturnal Hemoglobinuria (PNH)
- SLE
- Drugs
- Leukemia
- Hemophagocytic
- Lymphohistiocytosis (HLH)
Inherited Causes of Bone Marrow Failure
| Disease | Defect | Features |
| Fanconi anemia | Defect in DNA repair leads to increased chromosomal breakage with cross linking agents) | Skeletal anomalies (skeletal, thumb), short stature, urogenital anomalies |
| Dyskeratosis congenita | Shortened telomeres | Triad (leukoplakia, nail dystrophy, lacy skin pigmentation), pulmonary fibrosis |
| Shwachman Diamond syndrome | Mutation in SBDS gene | Exocrine pancreatic insufficiency |
| Congenital amegakaryocytic thrombocytopenia | Myeloproliferative Leukemia Virus Oncogene | Severe thrombocytopenia with its sequele |
| Hemophagocytic Lymphohistiocytosis | Multiple genes PFR1, UNC13D | Fever, splenomegaly, hepatitis, rash |
The acquired causes of pancytopenia
- noninherited aplastic anemia,
- megaloblastic anemia,
- malignant marrow infiltrative disorders (acute leukemias, myelodysplastic syndromes, solid tumors),
- non-malignant infiltrative disorders (storage disorders, osteopetriosis),
- infections(HIV, tuberculosis, hepatitis viruses, EBV, CMV),
- immune disorders (lupus, Evan’s syndrome, thymoma),
- acquired clonal bone marrow failure disorder (PNH),
- metabolic (anorexia nervosa)
- peripheral destruction of blood cells i.e. hypersplenism
Approach to Pancytopenia
Pancytopenia is always to be approached in a systematic manner through proper history, examination first and then moving on to laboratory investigations as the first two parts if done properly and a systematic manner will lead very near to the diagnosis.
History
The history should include the salient points viz
- age (inherited causes of bone marrow failure),
- sex,
- duration of symptoms (tells about the severity),
- bone pains (acute leukemias),
- fever (infections, acute leukemias),
- night sweats (Hodgkin’s disease),
- malaise, weight loss (tuberculosis, malignancy),
- bleeding from any site (magnitude of thrombocutopenia),
- jaundice (hepatitis viruses),
- joint pain, rash, photosensitivity (lupus),
- any radiation exposure,
- exposure to potentially toxic chemicals,
- treatment history including herbals and drug
- intake, blood transfusions, dietary history (megaloblastic anemia, anorexia nervosa),
- ooccupational exposure history (benzene).
Examination
A thorough examination should be done including
vitals, anthropometry, general physical examination and
systemic examination. Some salient features in
examination will point towards particular etiologies
like –
- anthropometry including stature (short stature in Fanconi anemia)
- dysmorphic features Fanconi anemia),
- pallor (severity of anemia),
- icterus (hepatitis viruses),
- lymphadenopathy (leukemias),
- edema, sings of CHF,
- stomatitis, cheilitis (neutropenia, neutrational deficiency),
- triad of nail dystrophy, leukoplakia, skin pigmentation (dyskeratosis congenita),
- oral candidiatis, pharyngeal exudates (neutropenia),
- petechie, purpura, hyperpigmentation, sternal tenderness(acuteleukemias),
- gum hypertrophy (Acute myeloid leukemia),
- hepatosplenomegaly, joint swelling, sinuvitis (lupus).
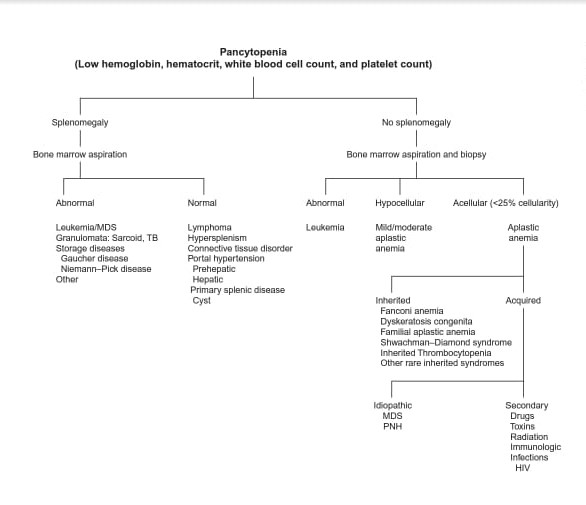
Pancytopenia with Splenomegaly :
Laboratory Investigation for Pancytopenia
- Complete blood counts (CBC) with RBC indices – Complete blood counts will show that all the 3 cell lines are decreased. Hb, Hct, MCV, ANC, Platelet count
- Peripheral blood smear (PBS) – Anisopoikiliocytosis i.e. variation in the sizes of RBCs alongwith the change in their shapes is very prominent in myelofibrosis whereas it is invariably absent in acute leukemias. Peripheral smears can show blasts (acute leukemias, myelofibrosis, subleukemic leukemias) or plasmacytic cells (multiple myeloma). RBC and WBC precursors in the peripheral blood are not typical of aplastic anemia, so their presence in pancytopenia suggests diagnosis other than aplastic anemia. Howel Jolly bodies (basophilic nuclear remnants) can be found in the peripheral smear of megaloblastic anemia or myelodysplastic syndrome. Hypersplenism and myelodysplastic syndrome peripheral smear can show giant platelets whereas normal sized platelets are found in aplasic anemia. The granulations of the neutrophils are to be taken into account; toxic granules are found in infections whereas hypogranulation of the neutrophils is a characteristic of myelodysplastic syndromes. The neutrophils can have hypersegmentaion (megaloblastic anemia) or hyposegmented (myelodysplastic syndromes, chronic leukemias).
- Reticulocyte count – In bone marrow failure where there is ineffective production of the cells by the marrow, the reticulocyte counts will be decreased, whereas it will be higher in cases where pancytopenia is secondary to the peripheral destruction of the cellular components of the blood.
- Bone marrow examination: aspiration and biopsy (to know the cellularity of the marrow) – Bone marrow examination is almost always indicated in cases of pancytopenia unless cause is very apparent. – Bone marrow aspirate permits examination of cytology (megaloblastic change, dysplastic change, abnormal cell infiltrate), immunophenotyping (leukemias, lymphoproliferative disorders) and cytogenetics (myelodysplasia, leukemias, lymphoproloferative disorders).
- Vit B12 and Folic Acid
- Viral Serology : Hepatitis A, B, C, EBV, parvovirus, varicella, CMV, HIV, PCR for Viruses when indicated
- Blood C/S : sepsis induced pancytopenia
- X-rays of the bones : multiple myeloma, metastasis
- Renal, Cardiac, Abdominal USG, Chest Xray – Congenital Anomalies
- LFT, KFT – etiology confining to these organs
- Chromosomal breakage analysis – FISH- diagnosing congenital bone marrow failure syndrome- Fanconi anemia
- Antinuclear Antibody (ANA Profile) – assay will point towards lupus
- Paroxysmal nocturnal hemoglobinuria (PNH Profile) –
The differentials of pancytopenia are based on the cellularity of the marrow as
- normocellular (50-70% hematopoietic cells with 30-50% fat),
- hypocellular (excessive amounts of fat cells)
- hypercellular (80- 100% cells with little fat);
| Bone Marrow Findings | Conditions |
| Cellularity | Hypercellular: Megaloblastic anemia, Hyperslenism Dry Tap: Myelofibrosis Hypoplastic: Myelodysplastic syndromes |
| Erythropoisis | Dysplastic: MDS, some AML Increased: Hemolysis |
| Myelopoisis | Dysplastic: Myelodysplastic syndrome Mophologically normal: Myeloproliferating disorders |
| Bone Marrow Findings | Conditions |
| Blasts | Myelodysplastic disorders, Acute Leukemias |
| Megakaryopoisis | Dysplastic: Myelodysplastic disorder |
| Other cells | Reedsternberg cell: Hodgkin cell Bacteria, Fungus, Parasite, Viruses, LD bodies |
Detailed Causes of Pancytopenia
Impaired production
Acquired aplastic anemia :
COMPLICATIONS
The major complications of severe pancytopenia are predominantly
related to the risk of life-threatening bleeding from prolonged thrombocytopenia or to infection secondary to protracted neutropenia. Patients
with protracted neutropenia as a result of bone marrow failure are at
risk not only for serious bacterial infections but also for invasive mycoses.
Patients who have been transfused with RBCs regularly over a long
period are at increased risk of developing alloantibodies to RBC antigens
and may require iron chelation therapy for transfusional iron overload.
The general principles of supportive care that have evolved from the
use of chemotherapy-related myelosuppression to treat patients with
cancer should be fully extended to the care of patients with acquired
pancytopenia.
PROGNOSIS
Spontaneous recovery from pancytopenia rarely occurs. If left untreated,
severe pancytopenia has an overall mortality rate of approximately 50%
within 6 mo of diagnosis and of >75% overall, with infection and
hemorrhage being the major causes of morbidity and mortality. The
majority of children with acquired severe aplastic anemia show response
to allogeneic marrow transplantation or immunosuppression, leaving
them with normal or near-normal blood cell counts.
Reference : https://www.ipinnovative.com/journal-article-file/3712